Page last updated: January 2025
The information on this webpage was adapted from Understanding Clinical Trials and Research - A guide for people with cancer, their families and friends (2025 edition). This webpage was last updated in January 2025.
Expert content reviewers:
All updated content has been clinically reviewed by
- A/Prof Brett Hughes, Senior Staff Specialist, Medical Oncology, Royal Brisbane and Women’s Hospital and The Prince Charles Hospital, and Associate Professor, The University of Queensland, QLD.
This edition is based on the previous edition, which was developed with help from a range of health professionals and people affected by cancer who have taken part in clinical trials. We thank the reviewers of this booklet:
- A/Prof Brett Hughes (see above)
- Christie Allan, Clinical Trials Lead, Cancer Council Victoria
- Dawn Bedwell, 13 11 20 Consultant, Cancer Council Queensland, QLD
- Joanne Benhamu, Senior Research Nurse, Team Lead, Lung, Colorectal and Palliative Care Trials, Parkville Cancer Clinical Trials Unit, Peter MacCallum Cancer Centre, VIC
- Louise Dillon, Consumer
- Sabina Jelinek, Clinical Nurse Research, St John of God Murdoch Hospital, WA
- Chloe Jennett, Program Coodinator, Cancer Research, Cancer Council NSW
- Carmel McCarthy, Consumer
- Alison Richards, Research Unit Manager, Medical Oncology Clinical Trials Unit, Flinders Medical Centre, SA
- Prof Jane Ussher, Translational Health Research Institute (THRI), School of Medicine, Western Sydney University, NSW
- Prof Janette Vardy, Medical Oncologist, Concord Cancer Centre, and Professor of Cancer Medicine, The University of Sydney, NSW
Clinical trials can help show whether a new approach to prevention, screening, diagnosis, or treatment works better than current methods and is safe.
People volunteer in clinical trials to help test how well the new way works and if it causes side effects or other problems.
If the new way is shown to work better than the existing method, it may become available. Some clinical trials compare existing approaches to see which one is more effective.
Search for clinical trials
Understand clinical trials in your language
Learn about the role and value of clinical trials through a series of multilingual videos produced by the Australian Clinical Trials Alliance.
The videos are available in Arabic, Chinese ( simplified and traditional), Filipino, Greek, Hindi, Italian, Punjabi, Spanish and Vietnamese. Written versions of the videos are also available.
More resources in other languages
Cancer research overview
Cancer research has led to the medical treatments and health programs available today. These advances have improved outcomes for people with all types of cancer over the past 20 years, with increases in both length of survival and quality of life.
The search for better ways to prevent, diagnose and treat cancer is ongoing. There are three main types of cancer research:
- population research – researchers known as epidemiologists look for patterns and trends to work out how and why cancers occur in groups of people (populations).
- laboratory research – scientists do experiments with the building blocks of disease, such as cells and blood, to try to understand how cancer works, and they also study and develop new drugs and treatments in the laboratory.
- clinical research – research is done on people to better diagnose, prevent and treat cancer, often in a hospital or treatment centre.
Population research and laboratory research are often the starting point for clinical research, which may involve clinical trials.
"I had to fill out a questionnaire about my emotional and physical wellbeing on the day of the trial and the following day. This gave me something to do and I felt that I was actively participating in my own health care" Marg
Cycle of research
The research process is a continuous cycle. People affected by cancer mainly take part in clinical trials, a type of clinical research. Research can lead to new and improved therapies becoming part of standard care.
- Population research identifies trend or problem (e.g. disease, risk factor)
- Laboratory research investigates a problem, develops a possible solution, and tests how well it works (in test tubes) and if it seems safe (in animals)
- Clinical research tests the solution (e.g. a new drug, treatment, program) on people
- New therapy may be approved for use on people if proven safe, effective and as good as or better than existing treatments; longterm benefits and risks will be monitored
Key questions
Why get involved in research?
When cancer patients, carers and survivors, as well as people not affected by cancer, take part in research, it helps researchers learn more about cancer and ways to treat and prevent it.
Many people diagnosed with cancer who join a clinical trial or another research study do so because they want to help improve outcomes for others in the future, as well as for themselves. Adults and children can take part in different ways, including:
- consenting to their medical records and personal information being accessed
- doing surveys and interviews
- being involved in a clinical trial
- agreeing to be examined regularly by health professionals
- allowing samples of cells or tissue taken during tests or treatment to be used for research outside of their own medical care.
Who can participate in research?
All research studies, including clinical trials, have guidelines setting out who can take part. These are known as eligibility criteria.
Most cancer research involves current patients, but some studies focus on cancer survivors, carers, family members, people at risk of cancer or people who have not been affected by cancer.
Anyone under the age of 18 needs permission from a parent or guardian before joining a research study.
To make sure results reflect Australia’s diverse population, it is important that research involves people of all ages, genders and sexualities, as well as people from a wide range of social, economic, racial and cultural backgrounds.
Where does research take place?
Cancer research is carried out in many places, including hospitals, treatment centres, laboratories and universities.
Sometimes you can be involved in cancer research from home. For example, you might have treatment or medicines mailed to you, or you might be asked to fill in a survey or complete a telephone or face-to-face interview.
In Australia, some people are now taking part in clinical trials that use telehealth (teletrials ).
Will I get better care in a clinical trial?
There are many advantages to being involved in a clinical trial or other research study. Depending on the type of research, the benefits may include:
- knowing you have made a valuable contribution to helping others in the future
- joining programs or having medicines or other treatments that are not readily available outside of the study and may be better than the current standard treatment for that disease
- getting access to expensive drugs that your specialists recommend but that are not currently supported by the Pharmaceutical Benefits Scheme (PBS)
- seeing your treatment team, including specialists, more often
- taking an active role in your health care
- learning new ways to improve your lifestyle
- improving or maintaining your quality of life
- feeling that you have tried all treatment possibilities.
Your doctor and the clinical trials or research nurse will discuss the possible advantages and disadvantages for you before you join a research study.
Taking part in research doesn’t always mean you will be better off than before or compared to other people in a similar situation. Some people may not respond in the way researchers hope and will not benefit from being involved in the research.
In some clinical trials, people are divided into two groups. Only one group receives the experimental treatment, while the other group receives the current standard treatment.
You won’t get to choose which treatment you have, but either way you will be monitored more frequently and closely than usual.
Is research safe?
Understandably, people want to know if there are any risks to taking part in research. Researchers must follow strict guidelines to make sure clinical trials and other research studies are as safe as possible for everyone involved. This is called their duty of care.
Before any research involving people can begin, it must be approved by a special group known as a human research ethics committee.
As part of this process, researchers identify risks that might occur, such as possible side effects. They must also explain how they will closely monitor these risks and what will be done if problems occur.
Before you agree to take part in research, you must be told about the risks, how you will be monitored for problems, and what will be done to help you if problems occur.
To reduce the risks, clinical trials are arranged in a series of steps known as phases.
How long do studies last?
From start to finish, clinical trials and other research studies may take several months or many years, but you may only need to be involved for some of this time.
For example, you might just have a single appointment lasting a couple of hours, or you may go to appointments every few weeks, months or years. You may also have to do surveys at regular times.
The participant information will set out what you would need to do and how long you would be involved.
Studies have what is known as a recruitment phase. This involves finding people to enrol in the study. It usually occurs over a few months or years until the required number of people have agreed to take part.
The study is then closed to new participants. After the treatment stage is over, there may be a follow-up phase. People may be followed up at set intervals for months or years.
This allows researchers to understand the long-term effects of treatments, monitor the general health of the participants, and collect data about long-term survival and quality of life.
Can I still have other treatments?
Ask your doctor whether being involved in the research will affect any other treatments you’re having or planning to have. These may include:
Your doctor may suggest stopping or delaying some treatments, or adjusting them in some way (e.g. by changing the dose).
It is important to let the research team know about any other medicines, supplements or complementary therapies you are having, as these may interact with the treatment being tested and cause harmful side effects.
Is it free to join a research study?
Joining a clinical trial or other research study is free for Australian citizens and residents, and you should not be asked to pay to join.
The cost of trial-related treatment, tests and check-ups will be paid for by the organisation that is funding or conducting the research, sometimes called the sponsor.
You will usually still have to pay for any treatments or tests you would normally pay for as part of your standard care. The participant information will outline any extra costs to you.
Will I be paid?
People participating in cancer research usually don’t get paid. This is because offering people money to join a clinical trial may put too much pressure on them to agree.
In some circumstances, you may be paid back for certain expenses (e.g. for travel, parking, light refreshments). These are known as out-of-pocket costs. The participant information will outline what expenses will be covered.
Can I be involved in more than one research study?
You may be interested in joining multiple clinical trials and other research studies. Check with the research team whether you can be part of more than one study at the same time.
If you can, think about whether you’ll be able to commit to all the requirements of the studies. With clinical trials for medicines, you can usually only join one trial at a time.
Who works on clinical trials?
A team of people work on research studies , and some of their roles may overlap. If you decide to join a clinical trial , you may have contact with all or some of these people:
- Investigator – also known as a researcher, an investigator develops and plans research studies, and obtains, analyses and publishes the results.
- Nurse or research assistant – coordinates finding people for the trial (recruitment) by talking to potential participants, making sure they are eligible and explaining the purpose of the trial, and acts as a link between the patient and the researchers or the health care team.
- Study coordinator – ensures the trial meets ethical and legal requirements, and applies for grants and manages budgets.
- Cancer specialist – may be a medical oncologist, surgeon, radiation oncologist or haematologist. A cancer specialist supervises your treatment, follow-up and overall care.
- Other professionals – including a pharmacist, allied health practitioners and complementary therapists.
In most cases, your cancer specialist will continue to look after your overall cancer care while you are on a clinical trial.
Teletrials for cancer
In Australia, some people are now taking part in teletrials.
A teletrial is where hospitals – regional, rural or city – share a clinical trial and work together to deliver it to their patients all at the same time. This means, for example, you may be on the same trial at your local hospital at the same time as someone 200km away at their local hospital.
The entire medical team involved in the one trial use digital communication, such as video conferencing or telehealth, to closely communicate with each other as one big team.
Teletrials were developed to make joining a clinical trial easier for people in rural and remote locations, but they can make it more convenient for anyone to take part.
You can talk to your cancer specialist about whether there is a teletrial you could join. If you are already involved in a clinical trial, you could ask if parts of it can be delivered by telehealth.
Learn more about teletrials
The clinical trials process
Cancer clinical trials are research studies that use volunteers to test new ways (interventions) to diagnose, treat and manage cancer.
If a trial proves that a test, treatment or other intervention is better than existing options, it may become the new standard of care for patients in the future.
A medicine or another intervention can be developed in a laboratory and tested on animals, but it is only once it has been tested on the human body that we can know it works in people.
It also has to be tested on enough people to show that any benefit is not just a random effect for a single person.
Types of trials
Clinical trials are designed to answer different research questions:
- Treatment trials – test new treatments, new ways of giving existing treatments, or new combinations of treatments. They look at whether the treatment works and if it causes side effects. The treatments that can be tested include: medicines (chemotherapy, immunotherapy, targeted therapy and other drugs); medical devices; radiation therapy; surgical techniques; nutrition advice; physiotherapy; exercise programs; psychological therapies; and complementary therapies. Most cancer clinical trials in Australia are treatment trials.
- Prevention trials – work out whether medicines and health programs lower the risk of developing diseases such as cancer.
- Screening trials – look at new approaches of detecting diseases before symptoms appear.
- Diagnostic trials – identify more accurate or easier tests for diagnosing a particular disease in people who have signs or symptoms.
- Quality of life trials – test ways to improve the comfort and quality of life of people who have cancer. They are done alongside a treatment trial.
“When I was diagnosed with cancer, a friend told me about the study. Getting involved was simple. Participating in cancer research is about giving to other people, and I think that’s a very valuable thing.” Phillipa
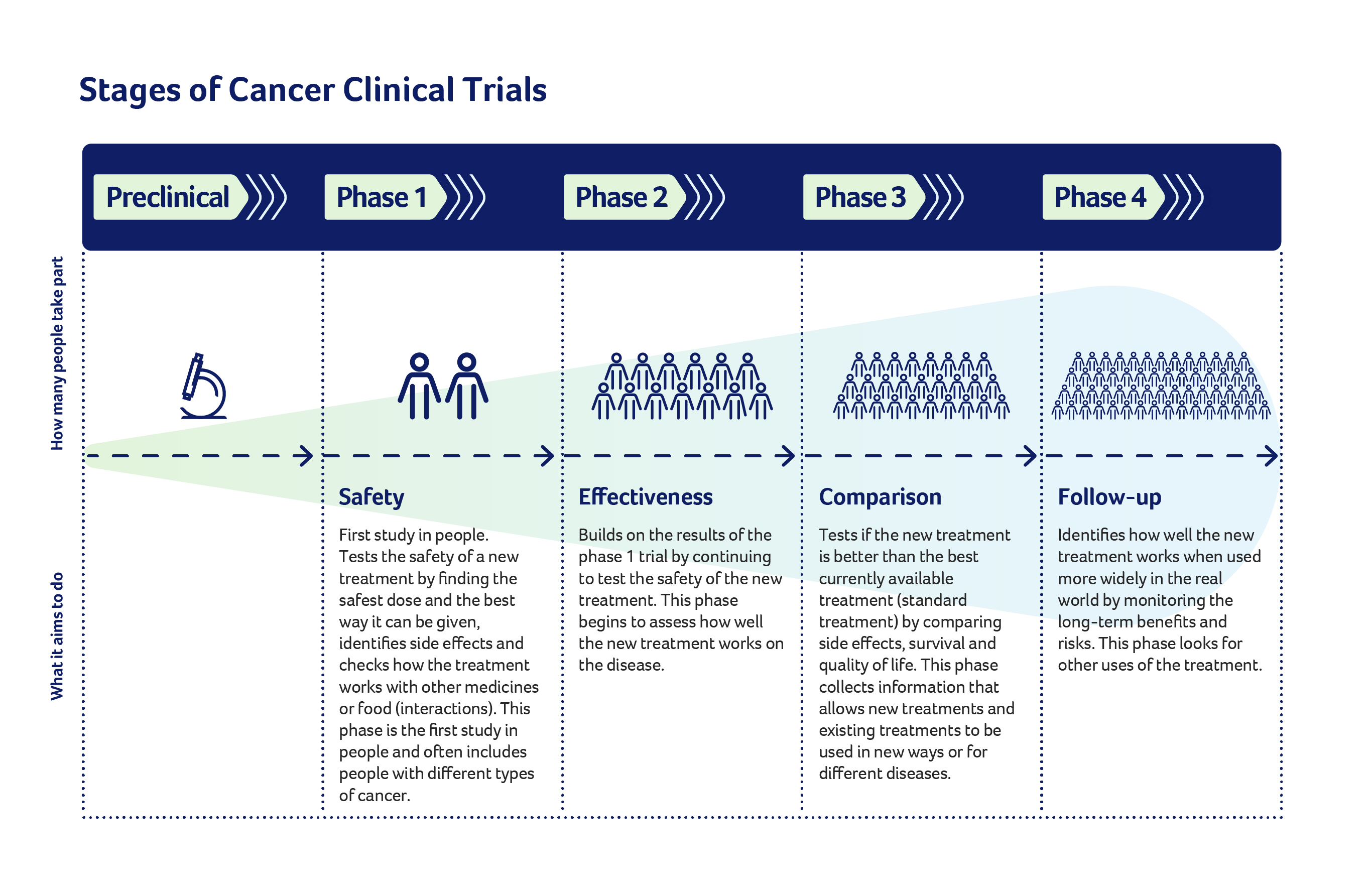
Phases of a clinical trial
Researchers spend many years developing new treatments or medicines in the laboratory before involving people.
They then plan the clinical trial to progress in a series of steps called phases. Every phase considers whether the risks outweigh the benefits.
Information gathered in each phase determines whether the study can move on to the next phase, and whether the drug or treatment is approved for use. There can be up to four phases, but not all clinical trials go through every phase.
Randomised controlled trials
It is important for researchers to know that the results of a study are accurate and not caused by chance. This means they must follow strict guidelines.
Researchers also need to make sure their own – or the participants’ – ideas or beliefs about the research don’t unfairly influence the results. There are various ways to make sure clinical trials are fair and reliable.
Many clinical trials are randomised controlled trials (RCTs). A randomised controlled trial helps prevent bias, so it is the best way to test if a new treatment works.
Bias occurs when the results of a trial are affected by human choice, expectations or other factors not related to the treatment being tested.
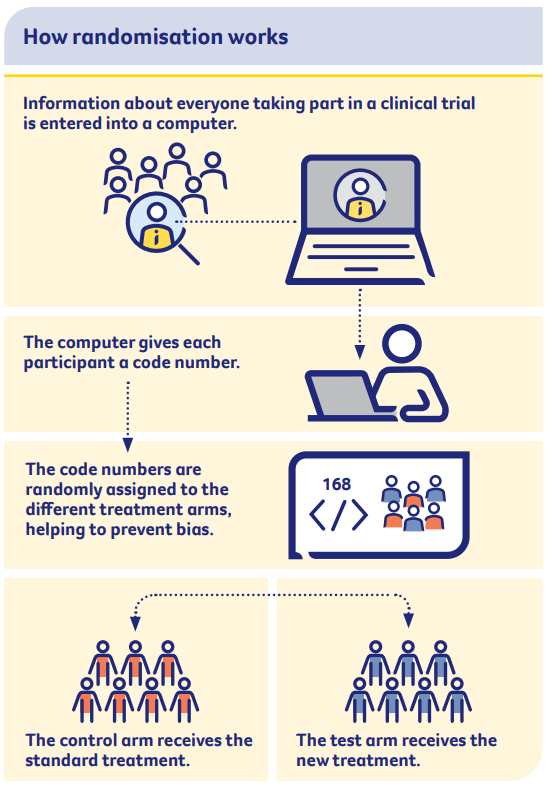
What is a randomised controlled trial?
Most phase 3 trials and some phase 2 trials are randomised. This means participants in the study are put into two or more groups (known as treatment arms) at random.
The two groups then receive different treatments, and the results of the different groups are compared. Researchers cannot choose who goes into each group.
- Test or experimental arm – this group is given the new treatment that is being tested. Sometimes, the experimental treatment is given in addition to the current standard treatment.
- Control arm – this group receives the current standard treatment for the disease or an inactive treatment known as a placebo.
When randomly allocated groups are compared with each other, it is possible to work out which treatment is better.
This is because researchers can be certain that the results are related to the treatment, and not to any other factors.
Standard treatment and placebos
In a randomised controlled trial, people in the control arm may receive the standard treatment or a placebo.
Standard treatment
This is the current most effective treatment given to people for their disease or condition.
For example, the standard treatment for early breast cancer is surgery, often followed by chemotherapy, radiation therapy and/or hormone therapy.
In some cases, the current standard of care is for the doctors to monitor the cancer closely with regular tests and check-ups, and only offer treatment if the cancer progresses.
This approach may be known as active surveillance or watchful waiting.
For example, active surveillance is recommended for some early thyroid cancers that aren’t causing any symptoms and are low risk, since the risks of treatment may outweigh the benefits in this situation.
Placebo
This is an inactive or “dummy” treatment. It is made to look, taste or feel like the treatment being tested, but it doesn’t have any active (therapeutic) ingredients (if a medicine) or beneficial effect.
Examples of placebos are sugar pills and saline injections.
A placebo is used to show whether any improvements are because of the actual treatment or because of other factors linked with being in the study, such as being more closely monitored or simply expecting the treatment to be helpful.
If the people on the experimental treatment improve more than those on the placebo, this provides stronger evidence that it is the experimental treatment that is responsible.
Participants will be told if a study uses a placebo, but they won’t be told which treatment they are having, and the research team usually don’t know either. In cancer treatment trials, placebos may be used:
- together with the standard treatment – for example, one group gets the existing standard therapy plus the experimental treatment, and the other group gets the standard treatment plus a placebo
- on their own, but only when there is no existing standard treatment to compare against an experimental treatment.
Blinded studies
In a blinded study, participants don’t know which arm of a study they’re in.
Some randomised controlled trials are called double-blind studies as neither the participant nor the research team know who is receiving the experimental or control treatment.
In a double-blind trial, the researchers only discover who is in each arm of the study at the end of the trial when the results are being analysed.
Blinding is used only when participants can’t tell the difference between the two types of treatment.
It is not used when the control and experimental treatment are noticeably different – for example, it would be hard to hide surgery and massage from the participant.
The aim of blinding is to reduce bias in the reporting of benefits and side effects. If you don’t know which treatment you’re having, the results are less likely to be influenced by your or your doctor’s views.
For example, if you or your doctor knew you were having the experimental treatment, you might report that you’re feeling better than you actually are because you believe you are having a more effective treatment.
In an emergency, your doctor can find out what treatment you’re having by contacting those running the study.
Crossover studies
In crossover studies, the people in each trial arm receive that treatment for a time and may then have the opportunity to swap to the other treatment.
This can mean all participants have all treatments and helps confirm which is the most effective. If the new treatment doesn’t work as hoped with the first group, the second group won’t cross over.
You also won’t cross over if you are doing well on the first treatment.
Non-randomised trials
In a single arm trial, everyone receives the same experimental treatment. This method may be used for phase 1 and phase 2 trials, or where the cancer being treated is rare and it is hard to conduct a randomised trial.
Registry trials
For most treatment trials, you will have the treatment as part of the trial. Another way researchers can compare how well different treatments work is through registry trials.
A registry trial collects information from people with a particular type of cancer who are having routine treatment.
It can’t be used for testing new treatments, but it can answer a range of questions about existing treatments. Your cancer treatment team may talk to you about joining a registry trial.
If you choose to join the registry, you agree to share your health information, such as your medical history, the treatments you are having and the results of tests.
This information will then be compared with information from many other patients to work out which treatment approaches are the most effective.
Registry trials are a cost-effective way to run a clinical trial and they allow many more people to participate in cancer research.
Other types of clinical research
Clinical trials are not the only type of clinical research studies. You may also be invited to get involved in behavioural research or translational research.
Behavioural research
Behavioural researchers try to understand why people behave in the way that they do. They study the characteristics, lifestyles and social situations of different people to see how these factors may affect the risk of developing or surviving cancer.
They then try to develop ways to prevent or change behaviours that might increase cancer risk or lower survival outcomes.
Behavioural researchers also look at the emotional and social impacts of cancer on the person with cancer, as well as the impact on their family and friends.
If you take part in a behavioural research study, you may be asked to fill in surveys or be interviewed about your lifestyle, including your eating, drinking, smoking and exercise habits.
You may also be able to take part in a program aimed at changing these behaviours. For example, you might be offered free counselling, an exercise class or a session on healthy eating.
The aim of the programs may be to reduce cancer risk or to improve how you cope with the impacts of cancer.
Psychosocial research
One area of behavioural research is called psychosocial research. This looks at how cancer affects people emotionally, psychologically and socially. In cancer care, this is sometimes called psycho-oncology.
Researchers try to understand how patients and carers cope emotionally at different stages of a disease. They develop and test ways to improve people’s ability to deal with the impacts of cancer.
Translational research
Translational research forms a link between laboratory and clinical research, or between clinical research and standard treatments. It aims to get new treatments or medical approaches into practice quickly.
It is sometimes called “bench to bedside” research because laboratory research results are directly used to create new therapies and tests.
The findings of behavioural and psychosocial research are also translated into information resources for people affected by cancer.
There can also be “bedside to bench” research – for example, hospitals, treatment centres and health professionals may collect information about how well a treatment works to help laboratory researchers work out what to study next.
Understanding Clinical Trials and Research
Download our Understanding Clinical Trials and Research booklet to learn more
Download now Order for free